“What Is RSV? CDC Warns of Respiratory Syncytial Virus Rising in the Southern U.S. - Prevention.com” plus 2 more
“What Is RSV? CDC Warns of Respiratory Syncytial Virus Rising in the Southern U.S. - Prevention.com” plus 2 more |
- What Is RSV? CDC Warns of Respiratory Syncytial Virus Rising in the Southern U.S. - Prevention.com
- SARS‐CoV‐2 and hypertension - Ravichandran - 2021 - Physiological Reports - Wiley Online Library - Wiley
- Does SARS-CoV-2 show seasonal variation in transmission in temperate countries? - News-Medical.Net
| What Is RSV? CDC Warns of Respiratory Syncytial Virus Rising in the Southern U.S. - Prevention.com Posted: 15 Jun 2021 12:10 PM PDT 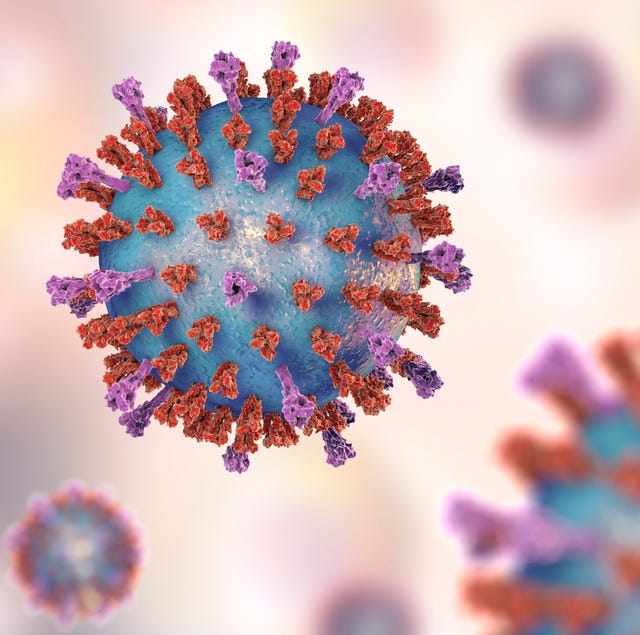 KATERYNA KON/SCIENCE PHOTO LIBRARYGetty Images
As COVID-19 infections continue to fall across the United States, experts are now warning of another disease that could threaten everyone from infants to the elderly: RSV, or respiratory syncytial virus. The Centers for Disease Control and Prevention (CDC) released a health advisory on June 10 about the rise of RSV, a respiratory illness that causes similar symptoms to COVID-19, in southern parts of the country. "Due to this increased activity, CDC encourages broader testing for RSV among patients presenting with acute respiratory illness who test negative for SARS-CoV-2, the virus that causes COVID-19," the advisory reads. Here's everything you need to know about respiratory syncytial virus, plus what you can to do to curb its spread. What is RSV, exactly?Respiratory syncytial virus is "a common respiratory virus that usually causes mild, cold-like symptoms," the CDC explains. Although most people recover in a week or two, RSV can cause severe illness, especially in infants and older people. Because it's a respiratory illness, RSV spreads much the same way COVID-19 does: Respiratory droplets from infected person (often through a cough or sneeze) makes contact with your eyes, nose or mouth or you touch a surface contaminated with the virus, then touch your face. Right now, the South is experiencing a sudden rise in RSV cases, which had been lower than normal since April 2020, the CDC notes. Lab-confirmed cases first started to rise in March 2021, and they've since skyrocketed in states like Florida, Georgia, North Carolina, Texas, and Oklahoma. What symptoms does RSV cause?RSV causes a few typical symptoms that are similar, but not identical, to those caused by COVID-19, per the CDC. These are the most common, which usually appear in stages—not all at once:
Most RSV infections clear up after two weeks. Patients who have trouble breathing or who are dehydrated should be hospitalized; in severe cases, a person might have to be put on oxygen or intubated, according to the CDC. Almost all children will have had an RSV infection by age 2, the CDC notes. Annually, 58,000 kids younger than five and 177,000 adults 65 or older are hospitalized with RSV; 100-500 of those children and 14,000 of those adults die of the disease. How is RSV treated?"There is no specific treatment for RSV infection," the CDC notes. Instead, patients can focus on easing their symptoms by taking over-the-counter fever reducers and pain relievers and drinking plenty of fluids. Parents should contact their child's pediatrician before administering nonprescription cold medicines, the CDC warns, because certain ingredients might be harmful to kids. You can prevent the spread of RSV by following the same precautions as you would with COVID-19, the CDC says, including washing your hands thoroughly and frequently, avoiding close contact with infected people, and wearing a mask or covering your sneezes and coughs if you're sick. If you suspect you have RSV (or have any cold-like symptoms), keep up with those practices and try to avoid close contact with children at high risk of severe illness, the CDC recommends. When in doubt, get tested. This content is created and maintained by a third party, and imported onto this page to help users provide their email addresses. You may be able to find more information about this and similar content at piano.io | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Posted: 13 Jun 2021 12:00 AM PDT 1 INTRODUCTIONHypertension (HT) is a serious public health problem affecting approximately 31.1% of adults worldwide. The disease is multifactorial and a risk factor for cardiovascular diseases, chronic kidney disease, stroke, and premature death (Appel et al., 2003; Mills et al., 2016). The classification of hypertension is given in Table 1.
The renin–angiotensin–aldosterone system (RAAS) plays an important role in regulating blood pressure (BP). BP regulation by the RAAS depends on the interplay of its two axes: angiotensin-converting enzyme/angiotensin-II/angiotensin type-I receptor (ACE/ANG-II/AT1R) and angiotensin-converting enzyme 2/angiotensin-(1-7)/MAS-receptor (ACE2/ANG-(1-7)/MAS). The ACE2/ANG-(1–7)/MAS-axis plays a protective role in several diseases, including HT (Azushima et al., 2020). The novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the cause of a worldwide pandemic. The virus appeared for the first time in Wuhan in China in 2019 and had been spreading ever since. The virus causes Coronavirus Disease 2019 (COVID-19) and Patients with COVID-19 exhibit various symptoms, such as among others fever and cough, but the disease can also lead to respiratory failure and death (Ludwig & Zarbock, 2020). Table 2 summarises several of known symptoms. As of 03/08/2021, 2.594.064 deaths related to COVID-19 were reported globally (Johns Hopkins University Coronavirus Resource Center, JHU, 2021). SARS-CoV-2 gains viral entry by interacting with membrane-bound angiotensin-converting-enzyme-2 (ACE2). The interaction may affect the expression of ACE2, which plays a key role in balancing the two axes of RAAS. (Azushima et al., 2020; Hoffmann et al., 2020; Li, He, et al., 2020).
The objective of this review is to summarise the pathophysiological effects of COVID-19 in relation to hypertension and with focus on the RAAS and the MAS receptor. 2 METHODSThe literature used for this review was mainly accessed through www.Pubmed.gov. Search terms were combined to limit the number of results. By filtering the results by year and only considering results starting from 2015, the number of hits were further decreased. Additionally, the number of results could be further reduced by searching for specific types of articles such as clinical studies, meta-analyses and clinical trials. 'Hypertension AND COVID-19' gave 56 results when filtering for meta-analysis'. 'COVID AND RAAS' gave 107 results. 'RAAS AND ACE2' gave 116 results. '2019-ncov AND structure' gave 120 results when filtering for clinical studies. '(RAAS +MAS receptor) OR (renin–angiotensin–aldosterone system +MAS receptor)' gave 426 results. 'COVID-19 AND vaccine' gave 36 results when filtering for clinical studies. Another way of finding articles was using the function 'Similar articles' in www. Pubmed.gov, and using the reference lists of other articles. Aside from www.Pubmed.gov, literature about treatment of hypertension, treatment of COVID-19 and routes of transmission were accessed through articles from www.who.int, and using the reference lists of the articles. Furthermore, some information about vaccines and drug candidates were accessed through www.ema.europa.eu. 3 HYPERTENSIONHypertension is a condition with a persistent elevated systolic (≥140 mmHg) (SBP) and/or diastolic blood pressure (DBP) (≥90 mmHg). Depending on the guidelines issued by the various societies, both the definition of hypertension and as the target blood pressure under therapy can vary. In 2020, the International Society of Hypertension has extracted the evidence-based content from multiple recent guidelines and proposed standards for worldwide practice, which can be applied both under low- as well as high-resource conditions. In general, BP is classified into four categories, as illustrated in Table 1 (Unger et al., 2020). According to the American Heart Association, HT is the main cause of death globally and compromises about 10.4 million people yearly. HT is a serious public health problem because it is a common risk factor for cardiovascular diseases like coronary artery disease (accounting for 25–30% of acute myocardial infarctions), stroke and heart failure (Unger et al., 2020). The disease is multifactorial, involving genetic and environmental factors such as smoking and diet (Rossier et al., 2017). There are two types of HT, primary and secondary HT. They are defined as HT with no clear underlying cause and HT with an underlying cause, respectively. 5–10% of HT patients have a secondary HT (Unger et al., 2020). The treatment of HT includes lifestyle modifications such as weight reduction and regular physical activity. Studies have shown that these behavioural changes lower BP and reduce the risk of cardiovascular diseases (Appel et al., 2003; Bacon et al., 2004; Unger et al., 2020). Patients with grade 1 and grade 2 HT are treated with antihypertensive drugs (Unger et al., 2020). The classes of blood pressure medications include among others AT1-Receptor antagonists (ARBs), ACE inhibitors (ACEIs), calcium channel blockers (CCBs), thiazides/thiazide-like diuretics, beta-adrenoreceptor antagonists (BAAs), α-adrenoreceptor antagonists, centrally active alpha-adrenergic agonists, alpha-2 receptor agonists and combined alpha- and beta-adrenoreceptor antagonists. First line drugs are RAAS blockers, CCBs and diuretics. Depending on the severity of HT, the drugs can be applied as mono- or combination therapy. The combination depends on the comorbidities or complication(s). Most patients with grade 1 or 2 HT require combination therapy (Unger et al., 2020; Williams et al., 2018). 4 THE RENIN–ANGIOTENSIN–ALDOSTERONE SYSTEM AND THE MAS RECEPTORThe RAAS is responsible for the regulation of BP, electrolyte balance and extracellular fluid. Dysregulation of the RAAS can lead to HT (Azushima et al., 2020; Patel et al., 2017). In response to a low BP, low NaCl and activity of the sympathetic nervous system, the renal juxtaglomerular cells release renin. This protein catalyses the formation of angiotensin I (ANG-I) from angiotensinogen, which is secreted from the liver. ANG-I is then cleaved by angiotensin-converting enzyme (ACE) producing angiotensin II (ANG-II). ANG-II binds to the ANG II type 1 receptor (AT1R) in various tissues in the body. The effects of the receptor activation are vasoconstriction, stimulation of aldosterone secretion, and stimulation of the thirst reflex leading to secretion of antidiuretic hormone (ADH). The released aldosterone stimulates the sodium and water reabsorption in the renal distal tubules and collecting ducts. ADH binds to receptors in the renal collecting ducts and decreases urinary loss. The effects of ANG-II, aldosterone and ADH increase cardiac pre- and afterload, leading to an increase in BP. Furthermore, the activation of the ACE/ANG-II/AT1R axis is linked to oxidative stress, fibrosis, and inflammation (Booth et al., 2002; Patel et al., 2017; Santos et al., 2019). ANG-II also binds to the ANG II type 2 receptor (AT2R). The effects of this interaction are the opposite of ANG-II activation of AT1R: antifibrosis, vasodilation, and anti-inflammation (Azushima et al., 2020). The ACE2/ANG-(1-7)/Mas axis is a counter-regulatory system of the ACE/ANG-II/AT1R axis; Figure 1 illustrates the two RAAS axes. The main regulatory enzyme in balancing the RAAS is ACE2, a membrane-bound monocarboxypeptidase expressed in cardiovascular tissue, the kidneys, lungs, liver, small intestine, and brain (Santos et al., 2019). It converts ANG-I to ANG-(1-9), but more importantly, it converts ANG-II to ANG-(1-7). Apart from having an affinity to the AT2R, ANG-(1-9) is cleaved by ACE producing ANG-(1-7). Thus, ACE2 reduces the amount of the substrate for ACE and the amount of circulating ANG-II, limiting the effects of the ACE/ANG-II/AT1R axis. ANG-(1-7) has a high affinity to the MAS receptor (MasR). The MasR is a G protein-coupled receptor. In humans it is mainly expressed in enterocytes, renal tubules, gallbladder, cardiomyocytes, male reproductive cells, placental trophoblasts, ductal cells, eye, and vasculature (Hikmet et al., 2020), and its activation leads to various effects such as antiarrhythmic effects and vasodilation through the release of nitric oxide (NO) and prostaglandins (Flores-Muñoz et al., 2011; Povlsen et al., 2020; Santos et al., 2013). Furthermore, studies on mice have shown that ANG-(1-7) reduces oxidative stress and vascular inflammation. Hammer et al. observed that a deletion of the MasR induced a proinflammatory phenotype of macrophages in mice (Hammer et al., 2016). In addition, Shenoy et al. observed that the ACE2/ANG-(1-7)/MAS axis is protecting the lungs from fibrosis and pulmonary hypertension in rats (Shenoy et al., 2010). These studies indicate that the ACE2/ANG-(1-7)/MAS axis has an anti-inflammatory and anti-fibrotic effect.  Overview of the RAAS. ANG-I, angiotensin I; ANG-II, angiotensin II; ANG-(1-7), angiotensin-(1-7); ANG-(1-9), Angiotensin-(1-9); ACE, angiotensin converting enzyme; ACE2, angiotensin converting enzyme 2; AT1R, angiotensin type-I receptor; AT2R, angiotensin type-II receptor, MasR, MAS receptor 5 SEVERE ACUTE RESPIRATORY SYNDROME CORONAVIRUS 2The novel SARS-CoV-2 is a spherical enveloped positive-strand RNA virus and a member of the β-coronavirus group (Zhu et al., 2020). It has a strong resemblance to SARS-CoV, sharing 80% of sequence identity and employing similar viral entry mechanisms (Chen et al., 2020). However, SARS-CoV-2 has a significantly higher binding affinity to ACE2, and thus, the number of virus particles required for infecting a cell is lower (Shang et al., 2020). The genome of SARS-CoV-2 codes for viral structural proteins such as spike (S) glycoprotein, envelope (E), membrane (M) and nucleocapsid (N). Especially the S protein plays a vital role in viral entry to the host cells, as illustrated in Figure 2 (Woo et al., 2010).  Schematic diagram of the RAAS, its protective effects on organs and its interactions with the SARS-CoV-2 viral entry mechanism. The protease renin cleaves angiotensinogen to generate angiotensin I. ACE plays an important role in converting angiotensin I into angiotensin II. Angiotensin II may exert some biological functions through angiotensin II receptor type 1 and 2 receptors (AT1R and AT2R), leading to potent vasoconstriction in several organs. ACE2 hydrolyses angiotensin II to the Angiotensin 1-7, which binds the Mas receptor playing a protective role in several organs. ACE2 also hydrolyses angiotensin I to the Angiotensin 1-9, which can be further converted to Angiotensin 1-7 by ACE. The balance between ACE/Ang II/AT1R and ACE2/Ang 1-7/MasR axes is a prerequisite for maintaining normal health. In addition, ADAM17 can cleave the extracellular juxta-membrane region of ACE2. Soluble ACE2 can bind to the receptor-binding domain of the spike protein of SARS-CoV-2 thus blocking further binding to ACE2 and host cell infection. (Small insert) Illustration of the viral entry of SARS-CoV-2. The receptor-binding domain of the spike protein from SARS-CoV-2 binds to ACE2, allowing host cell entry and infection. TMPRSS2: transmembrane protease serine 2. Adapted from (Abd El-Aziz et al. 2020) The interaction between the S protein, the serine protease transmembrane serine protease 2 (TMPRSS2) and membrane-bound ACE2 mediates the endocytosis of the virus into the cell. The S protein is a class I fusion protein and contains an S1 and S2 subunit. The S1 subunit mediates the attachment to membrane-bound ACE2. TMPRSS2 is co-expressed with ACE2 on type II pneumocytes. The enzyme induces conformational changes in the S2 subunit by cleavage. The conformational changes activate the subunit facilitating the membrane fusion into the host cell (Glowacka et al., 2011; Hoffmann et al., 2020; Wang, Zhang, Wu, et al., 2020; Zou et al., 2020). Apart from ACE2, alternative mechanisms of entry of the SARS-CoV-2 into the host cells are through interaction with AT2R and CD147. It has been shown that the AT2R has an affinity for the S protein and interacts with ACE2 and thus indicating the receptor playing a role in the viral entry into the host cells (Cui et al., 2020). In addition, the glycoprotein CD147 has been shown to interact with the S protein and facilitates viral entry to host cells suggesting CD147 as another possible receptor for viral entry of SARS-CoV-2 (Wang, Chen, et al., 2020). Following the endocytosis, the nucleocapsid is released into the cell where replication and translation of the genome occurs. The translated RNA and proteins are assembled in the endoplasmic reticulum and Golgi apparatus, after which the viruses are released from the cell by exocytosis (Hussain et al., 2020). The virus has a median incubation period of approximately 5 days, but it can last up to 14 days (Lauer et al., 2020). SARS-CoV-2 is transmitted from human to human (Li, Guan, et al., 2020). There have been cases with SARS-CoV-2 transmission from humans to animals such as dogs, cats, tigers, monkeys and minks. However, it is not clear if the virus can be transmitted back to humans again (Oreshkova et al., 2020; Sit et al., 2020). The main routes of transmission are through physical contact, respiratory droplets, fomites, and aerosols. The virus is capable of remaining infectious in aerosols and on surfaces for hours and days, respectively (van Doremalen et al., 2020; Li, Guan, et al., 2020; Tang et al., 2020). Some studies suggest that the virus may be transmitted faecal-orally (Chen, Chen, et al., 2020; Ong et al., 2020). There have also been cases indicating vertical transmission of the virus (Sisman et al., 2020; Vivanti et al., 2020). The carriers of SARS-CoV-2 can be either symptomatic, presymptomatic or asymptomatic (Furukawa et al., 2020; Li, Ji, et al., 2020; Rothe et al., 2020; Tong et al., 2020). Most patients infected with SARS-CoV-2 present mild symptoms such as fever and cough. Other symptoms that have been observed are malaise, dyspnoea, myalgia, sputum, fatigue, sore throat, and nausea (Du et al., 2020; Guan et al., 2020; da Rosa Mesquita et al., 2020; Wang, Yin, et al., 2020). In addition, a high prevalence of reversible olfactory and gustatory dysfunction was reported (Agyeman et al., 2020). Table 2 illustrates the prevalence of common symptoms of COVID-19. As a sign of worse disease progression, patients show leukopenia and lymphopenia (Guan et al., 2020; Yue et al., 2020). Chest computed tomography scans (CTs) of some patients with SARS-CoV-2 infection show bilateral pneumonia (Du et al., 2020; Guan et al., 2020; Wang, Yin, et al., 2020; Yue et al., 2020). Pneumonia is a common clinical manifestation of the COVID-19 and can lead to acute respiratory distress syndrome (ARDS). Other common complications are shock, acute kidney injury (AKI) and acute cardiac injury, with ARDS being the most common cause of death (Hussain et al., 2020; Wang, Yin, et al., 2020). Studies have also shown an increase in embolic events (pulmonary embolism, venous thromboembolic events, deep vein thrombosis) in patients with COVID-19 (Lu et al., 2020). Additionally, there have been cases with myocarditis, myopericarditis, acute haemorrhagic colitis and placental inflammation (Carvalho et al., 2020; Craver et al., 2020; Hosier et al., 2020; Kim et al., 2020; Naneishvili et al., 2020). Many patients that develop severe complications have comorbidities of HT, diabetes, or cardiovascular disease, indicating that these diseases might be risk factors for developing severe clinical manifestations (Du et al., 2020; Wang, Yin, et al., 2020; Zhou et al., 2020). Another significant risk factor for severe COVID-19 is an age over 65 years. Studies by Zhou et al. and Du et al. showed that the average age of nonsurvivors was 69 years, and 61% of 85 fatal cases were ≥65 years, respectively (Du et al., 2020; Zhou et al., 2020). Additionally, a meta-analysis by Zheng et al. estimated that patients with an age of >65 years have a 6-fold (OR = 6.01 (3.95, 9.16), p < 0.00001) increased risk of severe COVID-19 (Zheng et al., 2020). So far, approximately 2.593 million deaths related to COVID-19 have been reported (Johns Hopkins University Coronavirus Resource Center, March 8, 2021). The science regarding treatment of COVID-19 is fast-changing. Mainly symptomatic treatment is recommended by the WHO (WHO, 2020). Drugs such as hydroxychloroquine and remdesivir have been tested as treatments for patients with COVID-19. Remdesivir has been shown to have clinical benefits in relation to COVID-19 treatment, but a study has also found no significant benefits of the treatment. Also, hydroxychloroquine has shown not to be associated with clinical benefits (Beigel et al., 2020; Singh et al., 2020; Wang, Zhang, Du, et al., 2020). According to results from WHO's ongoing Solidarity Trial the antiviral therapies remdesivir, hydroxychloroquine, lopinavir/ritonavir and interferon had little or no effect on overall mortality in hospitalized patients with COVID-19, and only corticosteroids have been proven as an effective treatment against severe COVID-19 (Pan et al. 2020; The RECOVERY Collaborative Group (2021)). EMA has approved the use of the glucocorticoid dexamethasone for treatment of COVID-19 patients on oxygen or mechanical ventilation (EMA, 2020). A preliminary report shows that it can lower the 28-day mortality in this patient group (EMA, 2020; Horby et al., 2020). Additionally, FDA has authorized the monoclonal antibody therapies casirivimab and imdevimab for treatment of mild to moderate COVID-19 (FDA, 2020). Since it has been shown that COVID-19 increased the number of thrombotic events, heparin has been used to prevent these events. A study conducted in Spain showed that heparin correlated with a lower mortality (OR = 0.55 (0.37, 0.82), p = 0.003). Nevertheless, the evidence of this association is still limited, and further research is needed (Ayerbe et al., 2020). Human recombinant soluble ACE2 (hrsACE2) is a potential candidate for treatment of COVID-19. Using human blood vessels and kidney organoids, it has been shown to significantly inhibit SARS-Cov-2 infection in vitro (Monteil et al., 2020). Zoufaly el al. also reported that they saw promising results when they successfully treated a 45-year-old woman with severe COVID-19 with hrsACE2 therapy. The drug binds the viral S protein and thus neutralises SARS-CoV-2 and inhibits viral invasion into the host cells. Moreover, it converts ANG-II to ANG-(1-7) and ANG-I to ANG(1-9), thus upregulating the ACE2/ANG-(1-7)/MAS-axis (Zoufaly et al., 2020). Furthermore, due to downregulation of membrane-bound ACE2 in COVID-19 patients (Banu et al., 2020) and thus downregulation of the ACE2/ANG-(1-7)/MAS-axis, ANG-(1-7)-analogues are targets for therapy. Treatment of ANG-(1-7)-analogues could stimulate the anti-inflammatory and antifibrotic effects of the ACE2/ANG-(1-7)/MAS-axis. ANG-(1-7)-analogues are currently tested in clinical trials (NCT04332666, NCT04401423). Another candidate for drug targeting is the viral main protease Mpro, which plays a vital role in viral replication. Dai et al. and Ma et al. have developed inhibitors of Mpro which have shown promising results in vitro (Dai et al., 2020; Ma et al., 2020). Table 3 gives an overview of clinical studies involving RAAS-directed COVID-19 therapy approaches.
Currently, the availability of prophylaxis for COVID-19 is limited, but several vaccines are in preclinical and clinical trials worldwide. One vaccine has been approved for use in the Chinese military (Poland et al., 2020). The mRNA vaccine Gam-COVID-Vac (Sputnik V) has controversially also been approved for use in Russia (Callaway, 2020; Logunov et al., 2020). Pfizer and BioNTech announced the first results from a Phase 3 clinical trial of the vaccine candidate BNT162b2. The results show an efficacy rate above 90% in participants receiving a second dose of the vaccine (Polack et al., 2020). The vaccine has been approved for use in the United Kingdom, where vaccinations started on 8 December, 2020 (Hancock, 2020), as well as in the EU. Yet, persons with a history of a serious allergic reaction to a vaccine should not receive the vaccine, since there have been cases of anaphylactic reactions (Mahase, 2020). Furthermore, the Moderna and Astra Zeneca vaccines were also approved by EMA and FDA. Examples of COVID-19 vaccine candidates are listed in Table 4.
5.1 COVID-19: Effects on the RAAS and HypertensionA meta-analysis conducted by Zhang et al. showed an association between HT and COVID-19 severity and fatality. In patient groups with an age of <50 years and ≥50 years, the risk of severe HT was increased by a factor of 2.21 and 2.23, respectively. Additionally, the analysis showed that hypertensive patients have a 3.48-fold higher risk of fatality compared to nonhypertensive patients (Zhang, Wu, et al., 2020). Another meta-analysis by Pranata et al. included 6560 patients with COVID-19 pneumonia from 30 studies. The analysis showed that hypertension was associated with an increased severity of COVID-19 pneumonia (RR = 2.04 (1.69, 2.47), p <0.001) and increased mortality (RR 2.21 (1.74, 2.81), p <0.001) (Pranata et al., 2020). The studies imply that HT is a risk factor for severe or fatal COVID-19. The mechanism of HT in relation to severe COVID-19 is currently unclear. Since the virus enters the host cells by interacting with ACE2, it is hypothesised that the endocytosis of the virus-ACE2 complex reduces the function of ACE2, leading to an overactivation of the ACE/ANG-II/AT1R-axis and resulting in complications such as ARDS and multiorgan failure (Zhang, Wu, et al., 2020). Kuba et al. conducted an in vivo study on mouse lungs and showed a reduction of the ACE2 expression as a complication of the SARS-CoV infection. Besides, by blocking the RAAS pathway, they saw an attenuation of the effects of the SARS-CoV on the lungs (Kuba et al., 2005). Moreover, elevated levels of plasma-ANG-II were observed in patients suffering from COVID-19 (Liu et al., 2020). Plasma-ANG-II was linearly associated with viral load and lung injury (Liu et al., 2020). Additionally, studies have shown that patients with pulmonary arterial hypertension exhibited reduced ACE2 activity (Hemnes et al., 2018). These studies indicate that SARS-CoV-2 downregulates the expression of ACE2 on tissue, which leads to overactivity of the ACE/ANG-II/AT1R-axis and inhibition of the counterregulatory ACE2/ANG-(1-7)/MAS axis, resulting in lung injury. This can also explain why hypertensive patients are more prone to severe or fatal COVID-19 infections. One of the first-line medications in the treatment of HT are the blockers of the RAAS, ARBs and ACEIs (Williams et al., 2018). The effects of ARBs and ACEIs in relation to COVID-19 are highly debated. ACEI and ARBs induce an increase in ACE2 expression in cardiac tissue, and several tissues in rodents including cardiac and renal tissue. Therefore, some authors hypothesise that these drugs increase the risk of the SARS-CoV-2 infection. However, there is no significant evidence supporting the fact that ARBs and ACEIs increase the risk and severity of COVID-19 (Ferrario et al., 2005; Kreutz et al., 2020; Rico-Mesa et al., 2020). Furthermore, randomized clinical trials comparing the effects discontinuing vs. continuing ACEIs and ARBs therapy have found no evidence for discontinuing ARBs and ACEIs in patients with COVID-19 (Cohen et al. 2020; Lopes et al., 2021). On the contrary, it is more likely that the discontinuation of antihypertensive treatment increases the severity of COVID-19. Since the dysregulation of RAAS is a major factor in lung injury in COVID-19, the drugs might decrease the severity of symptoms by balancing the two axes of the RAAS. Zhang et al. conducted a retrospective study with 1128 HT-patients (ARB/ACEI-users and non-ARB/ACEI-users) infected with COVID-19. They found a significantly lower rate of mortality in the group with ARB/ACEI-users compared to the group of non-ARB/ACEI-users (adjusted HR = 0.42, p = 0.03). Likewise, they reported a lower incidence of septic shock and disseminated intravascular coagulation when comparing the two study groups (Zhang, Zhu, et al., 2020). Supporting this, Greco et al. performed a meta-analysis including 14 studies finding no increase in severe or fatal COVID-19 associated with an ARB or ACEI therapy (Greco et al., 2020). These studies illustrate the fact that rather than having a negative effect, the ARBs/ACEIs may have a protective effect in relation to the severity and fatality of COVID-19 in HT patients. Further studies are needed to determine the mechanisms behind the effect of the drugs in relation to COVID-19. The association between ARBs/ACEIs and risk of SARS-CoV-2 infection is yet to be determined. 6 DISCUSSIONThe COVID-19 pandemic is currently causing major health issues with a high rate of hospital admissions and deaths worldwide. Many patients are admitted to the hospital for a longer time and need an extended time to recover. Even patients who recovered from a moderate infection reported weakness, tiredness, and problems to smell and/or taste (Carfì et al., 2020; Rees et al., 2020; Tian et al., 2020). A recent meta-analysis also hinted towards a possible connection between COVID-19 and new-onset diabetes, showing that 14.4% of hospitalized COVID-19 patents also suffered from newly diagnosed diabetes (Sathish et al., 2020). Besides complications such as ARDS and AKI, SARS-CoV-2 also leads to cardiovascular damage (Craver et al., 2020; Kim et al., 2020; Lu et al., 2020; Naneishvili et al., 2020). Studies have shown the presence of SARS-CoV-2 genomes in cardiac tissue (Lindner et al., 2020). Furthermore, in endomyocardial biopsies permeation of small vascular walls was observed, and this is hypothesised to cause myocardial ischemia leading to arrythmias (Escher et al., 2020). SARS-CoV-2 has also been found within endothelial cells causing an endotheliitis, which is hypothesised to be the reason for the systemic complications of COVID-19. Therefore, stabilisation of vascular endothelium in combination with antiviral treatment could be an effective way of treating COVID-19 (Varga et al., 2020). It is obvious that HT is a major risk factor regarding the severity and fatality of COVID-19. However, it should be noted that the studies conducted by Zhang et al. and Pranata et al. exhibit some limitations. One limitation is the fact that the included studies can have different definitions of HT, leading to an overestimation or underestimation of the relation between HT and risk of severe COVID-19. Pranata et al. did also include many studies yet to be peer-reviewed. Lastly, information about the effect of antihypertensive medication could not be estimated, which underestimates the relation between HT and severity of COVID-19 (Pranata et al., 2020; Zhang, Wu, et al., 2020). Supporting the fact that HT increases the severity of COVID-19, it has been shown that treatment with ARBs or ACEIs decreases the risk of death in HT-patients with COVID-19 (Zhang, Zhu, et al., 2020). However, the number of patients receiving ARBs/ACEI in this study was only 188 which can cause statistical bias. Furthermore, the study is a retrospective study and not all data about the patients' medication could be retrieved (Zhang, Zhu, et al., 2020). However, a cohort study by Yan et al. illustrated a decreased fatality of COVID-19 patients with HT using ARBs. Weaknesses of this study are that the cohort is small (655 patients) and the majority were old patients (>65 years) (Yan et al., 2020). Hence, the study indicates protective effects of ARBs/ACEI regarding the treatment of hypertensive patients with severe COVID-19, but further studies are needed to confirm these effects. Most patients with severe COVID-19 have comorbidities, for instance, HT and diabetes, and they tend to be older (Du et al., 2020; Wang, Yin, et al., 2020; Zheng et al., 2020; Zhou et al., 2020). These patients are associated with ACE2 deficiency. Since studies indicate that ACE2 expression is reduced because of SARS-CoV-2 infection, this effect added to the ACE2 deficiency might worsen the dysregulation of the RAAS. This could explain the fact that patients with the mentioned features are more prone to severe COVID-19 (Verdecchia et al., 2020). Nonetheless, further studies are needed to clarify exactly why patients with features such as HT, diabetes and old age are more likely to have severe complications of COVID-19. Additionally, the mechanism of HT regarding severe COVID-19 should be determined. An infection of SARS-CoV-2 is associated with the decreased expression of ACE2 and elevated levels of plasma-ANG-II, indicating dysregulation of the two arms of RAAS (Kuba et al., 2005; Liu et al., 2020). Therefore, a potential target of COVID-19 treatment is increasing the plasma-levels of ACE2 through exogenous ACE2-therapy and thereby upregulating the ACE2/ANG-(1-7)/MAS-axis and downregulating the opposing axis of RAAS. Although there exists only a limited number of cases being treated with hrsACE2, the patients being treated have shown great results reinforcing the idea of future treatment of COVID-19 with exogenous ACE2 (Zoufaly et al., 2020). This review has some limitations. Firstly, there is publication bias since the references used the work were only in English, thus studies in other languages may have been excluded. Lastly, since the disease is relatively new, and most of the study population are Chinese, the geographic diversity is limited. 7 CONCLUSION AND OUTLOOKHT is a serious health problem worldwide, and studies have shown that it increases the severity and fatality of COVID-19. It has been demonstrated that RAAS plays a pivotal role in COVID-19 infection. It is known that an infection of SARS-CoV-2 can lead to a dysregulation of the RAAS by reducing the expression of ACE2. Although SARS-CoV-2 has emerged recently, the current treatments and the prophylaxis of COVID-19 is limited, although several drugs/vaccines as candidates for treatment/prophylaxis of COVID-19 have been tested in clinical trials. The research field is developing rapidly, and almost every day, new information and data about COVID-19 are published, such as a report on a SARS-CoV-2 blocker, which has fully prevented infection of cells in vitro (Svilenov et al., 2020) or on a ribonucleoside analogue, which was able to suppress SARS-CoV-2 transmission in ferrets (Cox et al., 2021). The exact mechanisms of HT in relation to COVID-19, and the biological processes behind the ACE2 downregulation due to SARS-CoV-2 remain uncertain, and further research is needed to determine these mechanisms. CONFLICTS OF INTERESTThe authors declare no conflict of interests. AUTHOR CONTRIBUTIONBriyanth Ravichandran, Daniela Grimm and Markus Wehland designed the present review. Briyanth Ravichandran and Marcus Krüger designed the figures. Briyanth Ravichandran, Daniela Grimm and Markus Wehland wrote the manuscript. All authors analyzed data and references. All authors read and approved the final manuscript. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Does SARS-CoV-2 show seasonal variation in transmission in temperate countries? - News-Medical.Net Posted: 15 Jun 2021 07:42 AM PDT The coronavirus disease 2019 (COVID-19) pandemic – caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) – was declared in March 2020, two months after the initial recognition of novel pneumonia-like symptoms in a group of patients in Wuhan, China. Since then, it has spread to over 192 countries and territories, causing well over 3.81 million deaths. The need to understand how and why such spread occurs is vital to containing it. A new medRxiv* preprint describes a significant reduction in transmission during the summer (by 42%), which is comparable to the impact of the most effective non-pharmaceutical intervention (NPI). -1.jpg) BackgroundMany respiratory infections, such as the influenza virus (or 'flu'), have been documented to have varying incidence rates or waves. Some earlier studies have suggested COVID-19 transmission can also vary with temperature, humidity, and ultraviolet radiation, while others denied such an association. These factors, related to climate and weather, are not stand-alones, however, since they mesh with many other biological and epidemiological factors to determine a pathogen's transmission dynamics. For instance, virus stability may vary with the weather and climate conditions, as may the level of susceptibility of the host. Humans also change their behavior with the seasons, staying home in winter but increasing their social and outdoor activity significantly with the onset of summer. Obviously, the latter will be associated with a much higher opportunity for viral transmission. Another factor is whether children are home, because of school holidays, or at school. Thus, even though the flu season occurs with "elegant and predictable periodicity" in any given set of years, the path to this regularity is not as obvious as if it were solely caused by seasonal variations in viral spread. As an earlier study on this topic has observed:
Examining time-dependent changes in transmissionFor this reason, the researchers focused on a single determinant of seasonality, in the form of the magnitude of variation in the time-dependent reproduction number, Rt, over a year, in the same climatic region. This avoids the difficulty of trying to tease out the individual and interrelated facets of seasonality. Importantly, the onset of the pandemic itself was marked by the implementation of NPIs of varying stringency, from national lockdowns and closure of international borders to travelers to face mask-wearing guidelines. Since these are hard on people and on the economy, they have been tightened and relaxed several times in succession over the 18 months since they were first imposed. Thus, the effect of NPIs on the seasonality of this pandemic must be accounted for in order to understand how seasonality drives the transmission of the virus. Decrease of 42% in summer transmissionThe scientists used data from older studies based in Europe alone to determine how environmental factors and behavioral changes were translated into seasonal effects. This showed that on July 1, the peak of summer, the reproduction number fell to below the peak of winter (on January 1), with the median decrease being 42%. This reduction was similar to that achieved by the most effective NPI even while falling short of that attained by combined measures, such as school and business closures; limiting of social gatherings to less than 10 to less than 1,000; and shelter-in-place mandates. Advantages of incorporating seasonality estimatesThese seasonality estimates held constant even when the data was confined to temperate countries in Europe. The study also helped understand how seasonality could help improve understanding of how effective NPIs really are by accounting for part of the residual variation in the Rt. For instance, in one model, a random walk was added on to the basic reproduction rate in order to dynamically explain transmission trends by attributing them to factors that were not included in the study. Such a random walk factor would shrink, becoming smaller and more uniform once seasonality is included in the reproduction rate estimates. Thus, the mean square displacement (MSD) of the random walk is 0.131 log and 0.072 log when applied to the non-seasonal and seasonal estimate, respectively. This means that quite a bit of the residual variation is attributable to the seasonality in both cases. These estimates remain robust when the reduction in transmission on any given peak seasonality day in December or January is calculated, with less than 5% difference between peak or trough estimates. What are the implications?The results of this study indicate that the peak transmission rates of SARS-CoV-2 in summer is about 42% lower, relative to the winter peak, attributable solely to seasonality. The findings correlate with earlier assumptions of 10% to 40% declines in the rate of transmission of this virus from winter to summer peaks. Resurgences may happen in colder regions in summer as well as in winter, however, as well as in warmer countries, with the Rt remaining above 1. It is also important to remember that latitude changes a whole array of population-based factors, sociocultural and epidemiological attributes, and thus the transmission dynamics. Closer attention must be paid to separate aspects of seasonality, especially weather and climatic conditions vs. behavioral changes, the latter being very unique during 2020 and unlikely to operate again in the near future. Yet, the single estimate of seasonality magnitude may be enough to provide a picture of what happens over time, and to strategize accordingly, even without adequate knowledge of each separate factor. Its ability to provide a more dependable idea of what may be expected in terms of transmission is likely to be important in achieving more accurate models, especially when accounting for the effects of NPIs. Considering that school holidays are responsible for a large part of seasonal variations in other respiratory viral diseases, the fact that they are included only as NPIs in this model underlines the importance of the observed seasonality of SARS-CoV-2. Since population immunity was being built over the first year of the pandemic, it seems that "going forward, the long-term seasonality of SARS-CoV-2 will depend in part on developments in population immunity as well as on the emergence of variants." In short, seasonality may help understand the reduction in COVID-19 transmission in summer apart from population-level immunity, thus promoting proper preparation for a fall or winter surge. Secondly, knowing the limits of the role played by weather and climate factors will help prevent over-dependence on seasonal variation and encourage NPI strategies to contain viral spread. *Important noticemedRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information. Journal reference: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| You are subscribed to email updates from "what can cause respiratory distress,what causes sars,what is acute respiratory infection" - Google News. To stop receiving these emails, you may unsubscribe now. | Email delivery powered by Google |
| Google, 1600 Amphitheatre Parkway, Mountain View, CA 94043, United States | |
Comments
Post a Comment